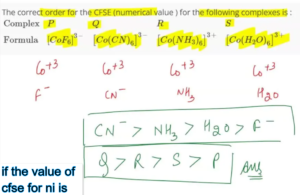
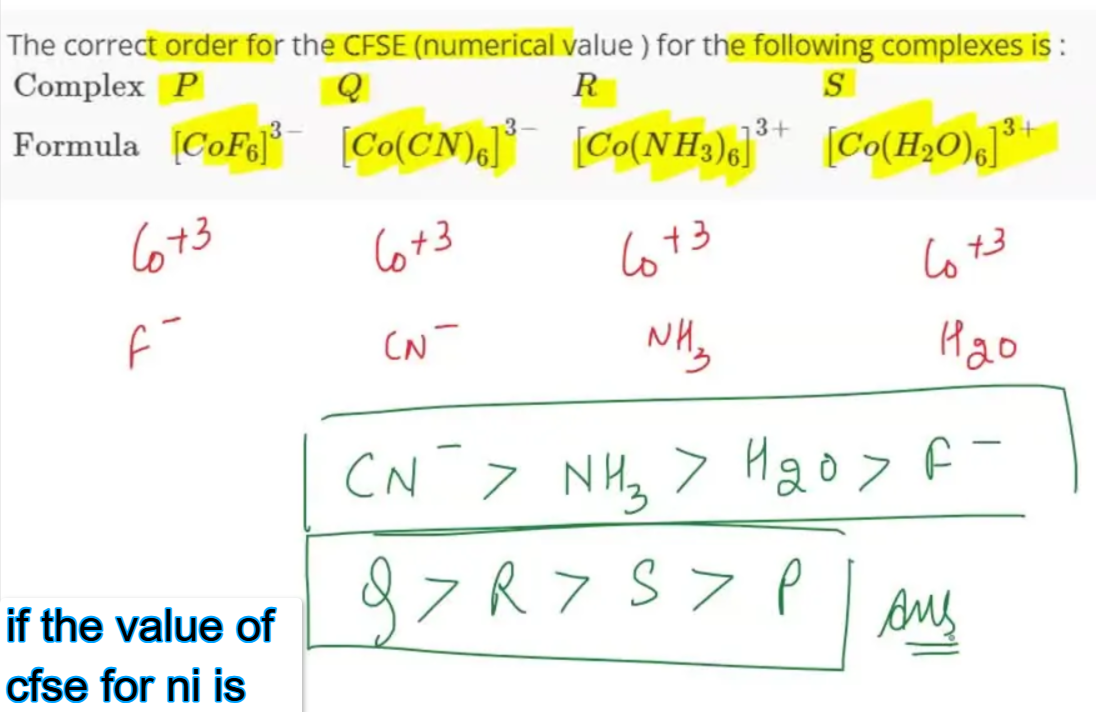
Crystal Field Stabilization Energy (CFSE) is a fundamental notion in coordination science. It helps explain the stability and magnetism of color, color and the structure of Transition Metal Complexes. Nickel (Ni) is a transition metal, with partially filled d-orbitals forms numerous complexes that can be understood through the theory of crystal fields. To understand the characteristics, chemists usually study how the d-orbitals divide between different ligand environments and how electrons interact with these. In this article we will examine the significance and implications for CFSE for nickel-based complexes. We will also investigate what happens if the value of cfse for ni is examined under various geometries of coordination.
The crystal field theory the five degenerate orbitals of an ion of transition metal split into groups with different energies when enclosed by the ligands. The manner in which they split is dependent on the structure that the complex is, which is most typically octahedral or trihedral. In an octahedral area the d-orbitals break up into two distinct groups: lower energy orbitals t2g and the higher energy e_g orbitals. Electrons are able to fill these orbitals in accordance with Hund’s rule as well as that is, the Pauli exclusion principle and the Aufbau principle.
Nickel typically forms the Ni2+ Ion in complexes. The electrochemical configuration of nickel neutral is [Ar] 3d8 4s2 and when it is formed into Ni2+ it sheds 2 electrons in the orbital 4s which results in an arrangement of 3d8. In an octahedral compound, the eight d-electrons are scattered across the split orbitals in t2g6 and e_g2. Since electrons in orbitals of t2g are responsible for stabilization, while those in the e_g orbitals are responsible for destabilization, CFSE could be calculated from these electron placements.
For an octahedral complex of Ni2+ The CFSE can be calculated by formula: CFSE = (-0.4 x the number of electrons in the t2g) + (0.6 x the number of electrons within e_g) multiplied by D0 (the Octahedral Splitting Energy).
Substituting the values of Ni2+ results in:
CFSE = (-0.4 x 6 + 0.6 x 2)D0
= (-2.4 + 1.2)D0
= -1.2D0
The negative value means that the complex is stable in comparison to the hypothetical split orbital configuration. Chemists who study stability of complexes they usually pose questions such as if the value of cfse for ni is -1.2D0 for an octahedral space what is the relationship with tetrahedral and square-planar configurations. The answer can help determine which geometry is the most suitable.
In tetrahedral compounds, the pattern of splitting is reversed, and the split pattern is smaller in terms of magnitude, which results in distinct CFSE values. However, a lot of nickel(II) complexes particularly those with strong field ligands use a square-planar geometry that results in even more stability for the d8 configuration. This is the reason that complexes like [Ni(CN)4]2are square-planar and diamagnetic.
Knowing CFSE within nickel complexes are crucial in areas such as catalysis, inorganic chemistry as well as materials sciences. By studying the process of orbital splitting and the energy of stabilization, scientists can determine the magnetic properties and reactivity of the metal complexes. In the end, studying situations in which if the value of cfse for ni is is established, scientists can discover which structures are energetically advantageous and the reason what causes certain nickel complexes to develop more quickly than others.